NOVAC Marian1, OZON Emma Adriana1, LUPULEASA Dumitru1, MITITELU Magdalena2*, NICOLESCU Teodor Octavian3, NICOLESCU Florica4, IONIȚĂ Elena Iuliana5, NEACȘU Sorinel Marius2, GHICA Manuela6, DUMITRESCU Denisa Elena7
1Pharmaceutical Technology Department, Faculty of Pharmacy, ”Carol Davila” University of Medicine and Pharmacy, 6, Traian Vuia Street, 020956, Bucharest (ROMANIA)
2Clinical Laboratory and Food Hygiene Department, Faculty of Pharmacy, ”Carol Davila” University of Medicine and Pharmacy, 6, Traian Vuia Street, 020956, Bucharest (ROMANIA)
3 Organic Chemistry Department, Faculty of Pharmacy, ”Carol Davila” University of Medicine and Pharmacy, 6, Traian Vuia Street, 020956, Bucharest (ROMANIA)
4Toxicology Department, Faculty of Pharmacy, ”Carol Davila” University of Medicine and Pharmacy, 6, Traian Vuia Street, 020956, Bucharest (ROMANIA)
5Pharmacognosy, Phytochemistry, Phytotherapy Department, Faculty of Pharmacy, ”Carol Davila” University of Medicine and Pharmacy, 6, Traian Vuia Street, 020956, Bucharest (ROMANIA)
6Department of Mathematics and Biostatistics, Faculty of Pharmacy, ”Carol Davila” University of Medicine and Pharmacy, 6, Traian Vuia Street, 020956, Bucharest (ROMANIA)
7 Organic Chemistry Department, Faculty of Pharmacy, Ovidius University, Constanța (ROMANIA)
*corresponding author: mititelumagdalena@yahoo.com
Abstract
In recent years, cyclodextrins (CDs) have been one of the topics of great interest to many industries, including the pharmaceutical industry. These are due to their particular structure which gives them interesting physicochemical properties, one of the most important being their ability to form inclusion complexes with a wide variety of molecules, compounds that are more soluble than the active substance as such, increasing them thus bioavailability. It should be noted that with the improvement of bioavailability, an improved stability of the active substances in solid, semi-solid and even liquid formulations is also obtained.
Keywords: cyclodextrins, bioavailability, stability, pharmaceutical industry
Introduction
Cyclodextrins are a family of starch-derived cyclic oligosaccharides. In 1891, Villiers discovered crystalline dextrins (“cellulosics”) in the bacterial digestion of starch; they have a cyclic structure and are produced by a bacillus (Bacillus macerans). The structures of the first cyclic compounds, respectively α – β – γ – cyclodextrins, were elucidated in 1942 and 1948, the detailed structure of δ – cyclodextrin was published in 1990. About 15 bacterial species producing CGTases are currently known. They can be classified into α – β – γ – CGT – aze, depending on the type of cyclodextrin initially obtained [1,3,4].
In 1981, the first International Symposium on Cyclodextrins was held in Budapest, during which Szejtli, one of the pioneers of the use of cyclodextrins, planned an industrial-scale production of these compounds at a reasonable price, as well as their use in the food industry. pharmaceutical.
The use of cyclodextrins in various fields (food, chemical, pharmaceutical, etc.) has been authorized since the 1980s. Thus, the Ministries of Health of Japan (1983) and France (1986) approved, based on metabolic and toxicological studies, the use of β – cyclodextrin for food flavor stabilization, the Swiss Ministries of Health (1986), Belgium, Luxembourg, Spain, have declared cyclodextrins to be usable in all foods that allow the use of enzymatically modified starch. In Germany alone, β-cyclodextrins are considered to be non-toxic auxiliaries for drug preparation [4,5,6].
Cyclodextrins (CDs) are cyclomaltooligosallarides consisting of six, seven or eight units of D-glucopyranose, (α-β-γ-cyclodextrin). Their structure is shown in Figure 1.
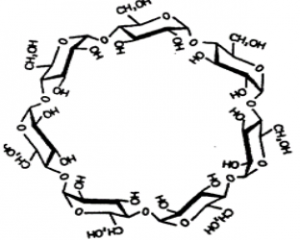
Fig.1 The structure of β – cyclodextrin [1]
Preparation:
CDs are obtained by an enzymatic reaction, starting from starch, by means of cyclodextrin – glycosyltransferase (CGT-ase – E.C. 2.4.1.19):
![]()
The microorganisms used for the production of CGT-ases are: Bacillus circulans, Bacitlus stearotermophilus, Bacillus megaterium, Bacillus licheniformis, Bacillus ohbensis, Micrococcus variants, Klebsiella pneumoniae, Bacillus macerans. Cyclodextrin-glycosyltransferases produce α, β, γ -cyclodextrins starting from starch, but the composition of the obtained cyclodextrins is different, depending on the enzymes used [2,3].
The main reaction product of all CGT-ases is β-cyclodextrin.
The disadvantages of this preparation method are:
-pretreatment or liquefaction of starch with the help of α –amylase for the solubilization of starch; after liquefaction, α -amylase must inactivated to obtain a good yield in cyclodextrins;
-cyclodextrin-glycosyltransferases are not thermostable enough to be used at high temperatures, so the conversion of starch cyclodextrins require a long reaction time for achieving a reasonable yield [2,3].
In the late 1960s, Armbruster perfected the technique of producing α-cyclodextrin. It develops a very efficient process, by using complexing agents for α-cyclodextrin, for example 1-decanol. Today, there are only a few companies that produce α – cyclodextrin [1,2,3].
Regarding γ – cyclodextrin, it is also formed by the action of CGT – axes, but in limited quantities; obtaining γ-cyclodextrin in larger quantities can only be achieved using a complexing agent. Horikoshi and colleagues develop a process for obtaining γ-cyclodextrin using organic complexing agents. Szejtli et al. are developing a process based on the concomitant use of two complexing agents, for example methyl ethyl ketone and α-naphthol [1,2].
Hans Bender similarly obtains γ-cyclodextrin by using bromobenzene as a complexing agent. Sato et al. publish a process for obtaining γ-cyclodextrin using tetra- or pentacyclic triterpenes (eg, glycyrrhizin). These complexing agents are selective for γ-cyclodextrin, the the yield being much higher than in the case of the aforementioned procedures [2,3].
Physical – chemical characteristics
CDs are crystallized non-reducing cyclic oligosaccharides also known as: cycloglucans, cyclomaltoligosides, SCHARDINGER dextrins that have the ability to form inclusion complexes with a wide range of hardly water-soluble medicinal substances. The best known and studied α, β, γ are – CD corresponding to: cyclohexaamylosis, cycloheptaamylose, cyclooctaamylose [2,3,4].
As we have shown, cyclodextrins consist of 6, 7 and 8 units of glucose (pyranose form), linked in the form of an empty cylinder, of molecular size (fig.2).

Fig. 2. Molecular dimensions of α, β, and γ – cyclodextrins (Bender M. L. – 1978)
All secondary -OH groups are located on the edge, and the primary -OH groups are on either side of the ring so that on the outside the CD molecule is hydrophilic. Due to this particular arrangement of glucose units, the interior of the CD ring consists of two rings of C-H groups and one ring of oxygen groups (O) included in glycosidic bonds. The free oxygen electrons are directed to the inside of the CD cavity, giving it an apolar character [3,4,5].
In the hollow annular space of these bonds which have a limited volume (outer diameter of a cyclodextrin cylinder having values of 1.4 – 1.7 nm) can be included only molecules or functional groups of molecules less hydrophilic than water, so with a suitable structure with a limited molecular size, compatible with that space. Many molecules are able to enter the cyclodextrin cavity, thus forming an inclusion compound. These molecules must satisfy a major condition, namely the so-called ability to adapt totally or partially to the cyclodextrin cavity; β and γ -cyclodextrins are the most convenient for pharmaceutical technology, because the size of their cavity (Φ = 6.0 ÷ 6.4 A and Φ = 7.5 ÷ 8.3 A, respectively) allows the inclusion of drug molecules, α -cyclodextrin is generally too small for this purpose [3,5,7].
Cyclodextrins are readily soluble in water, with β-cyclodextrin being the least soluble (1.8 g / 100 ml). The solubility of cyclodextrins increases with increasing temperature; upon cooling a rapid recrystallization takes place [3,7].
By various chemical modifications, semisynthetic derivatives of β -cyclodextrin were obtained which were divided into three groups: hydrophilic, hydrophobic and ionic.
Hydrophilic derivatives remarkably improve the solubility, chemical stability and bioavailability of many drugs. The hydrophilic derivatives of β -cyclodextrin are in turn divided into three groups: methylated, hydroxyalkylated and branched. The water solubility of β-cyclodextrin is significantly increased by the introduction of methyl, hydroxyalkyl or glucosyl groups [3,7].
The metabolism of cyclodextrins takes place at a lower initial rate than that of starch. This delay in cyclodextrin metabolism can be explained by the fact that cyclodextrins are totally resistant to the enzymatic degradation of terminal groups, so that they are not hydrolyzed by α 1,4-glucanoidases, as fast as linear dextrins. This has been demonstrated by studies in mice, in which the maximum amount of expired CO occurs after one hour in the case of starch and only after 9 hours in the case of cyclodextrins. The metabolism of decomposed cyclodextrins is the same as in starch [2,3,7].
Long-term carcinogenicity studies (2 years) performed in mice dieting with 3 ÷ 6% β -cyclodextrin did not indicate carcinogenic effects. Also, “in vivo” mutagenicity studies did not reveal mutagenic properties. Among the derivatives of natural cyclodextrins, the hydrophilic derivatives of β-cyclodextrin enjoy maximum attention [2,8,9,10,11].
As shown, DM-β-CD dimethylated derivative of β-cyclodextrin is more soluble in water than β-cyclodextrin and forms with lipophilic compounds, in general, more stable inclusion compounds than β-cyclodextrin. DM-β-CD is also more lipophilic than β-cyclodextrin and as a result there is a detergent-like effect on biological membranes that leads to significant hemolysis both “in vitro” and “in vivo“. These physical properties give this derivative a low lethal dose (LD) (200 mg / kg) for parenteral administration, which excludes its use in formulations used for the i.v. [12-16]
For 2-HP-β-CD, the major hydroxyalkylated derivative of β-cyclodextrin, studies in healthy male and female volunteers outlined the following pharmacokinetic picture: after oral administration, regardless of the dose applied, its presence in plasma or urine could not be determined, indicating a lack of absorption from the gastrointestinal tract [17,21].
Conclusions
Cyclodextrins are used as “excipients” in various other industries. In the chemical industry it is used for:
– obtaining herbicides, flavorings and dyes;
– initiation of catalytic reactions;
– separation of enantiomers by gas chromatography (GC) and high performance liquid chromatography (HPLC).
They are also used extensively as additives in the food and cosmetics industries.
Many volatile products such as volatile oils, menthol, camphor, anethole have been included in cyclodextrins. The inclusion compounds obtained facilitate the easy handling of the products due to the fact that they are in a solid state. Moreover, the volatility of substances is considerably reduced.
Due to the complexity of their composition, not all volatile oil molecules have the ability to be equally included in a given cyclodextrin. In all cases, however, the value of these inclusions must lead to an improvement in the quality of the pharmaceutical forms in which they are incorporated, in particular suppositories and inhalations. The formation of the inclusion compounds increases the boiling point of a liquid starting product and can increase the melting point and sublimation temperature of an initially solid substance.
The inclusion of a substance in the CD can improve oxidation resistance. This has been shown for vitamins, especially for vitamin D3. Thus, the inclusion compound vitamin D3: β -CD fixes only 11.2% of the fixed amount of oxygen of pure vitamin over the same experimental period of time [22,23].
Improving the stability of CD inclusion is not as safe in terms of hydrolysis. In this case, the stability depends mainly on the nature of the substance included, the type of CD and the pH of the medium. No definitive conclusions can be drawn at this time [23].
In topical preparations where there is intimate contact between the active substance and the excipient, the inclusion of the active substance may improve its stability, as demonstrated by numerous data in the literature. Thus, studies with included and non-included corticosteroids incorporated in various ointment bases, showed that after one month, the loss of active substance is in the first case 5%, and in the second case about 40 ÷ 50% [4,5,22].
REFERENCES
- Wimmer, T., (2012). Cyclodextrins. Ullmann’s Encyclopedia of Industrial Chemistry. Wiley-VCH
- Szejtli, J. (1998). “Introduction and General Overview of Cyclodextrin Chemistry”. Chem. Rev. 98 (5): 1743–1754
- Biwer, A., Antranikian, G., Heinzle, E. (2002). Enzymatic production of cyclodextrins. Applied Microbiology and Biotechnology. 59 (6): 609–17
- Uekama, K., Hirayama, F., Irie, T. (1998). Cyclodextrin Drug Carrier Systems. Chemical Reviews. 98 (5): 2045–2076
- Bender, H. (1986). Production, characterization and application of cyclodextrins, Adv.Biotechnol.Prousses, p.631 – 671
- Gao, B., Wang, G., Li, B., Wu, L. (2020). Self-Inclusion and Dissociation of a Bridging β-Cyclodextrin Triplet. ACS Omega, 5 (14) , 8127-8136
- Becket, G., Schep, L. J., Tan, M. Y. (1999). “Improvement of the in vitro dissolution of praziquantel by complexation with α-, β- and γ-cyclodextrins”. International Journal of Pharmaceutics. 179 (1): 65–71
- Drăgoi, C. M., Moroșan, E., Dumitrescu, I. B., Nicolae, A. C., Arsene, A. L., Drăgănescu, D., Lupuliasa, D., Ioniță, A. C., Pantea A., Stoian, C. Nicolae, Rizzo, M., Mititelu, M. (2019). Insights into chrononutrition: the innermost interplay amongst nutrition, metabolism and the circadian clock, in the context of epigenetic reprogramming. Farmacia 67(4), pp. 557-571
- Ioniță, A. C., Ghica, M., Moroșan, E., Nicolescu, F., Mititelu, M. (2019). In vitro effects of some synthesized aminoacetanilides N’- substituted on human leukocytes separated from peripheral blood. Farmacia 67(4), pp. 684-690
- Ioniță, A. C., Nicolescu, T. O., Ioniţă, E. I., Nicolescu, F., Mititelu, M. (2020). Chemical-Toxicological Research on the Evaluation of Serum Malondialdehyde Levels in Patients with Diabetic Arteriopathy. Proceedings of 6 th International Conference on Interdisciplinary Management of Diabetes Mellitus and its Complications INTERDIAB (Bucharest, Romania, 5-7 March 2020), Filodiritto Editore – Proceedings, pp. 320-325
- Mititelu, M., Nicolescu, T. O., Udeanu, D. I., Nicolescu, F. (2020). Research on the Influence of Some Sweeteners on Body Weight and Blood Glucose in Laboratory Mice. Proceedings of 6 th International Conference on Interdisciplinary Management of Diabetes Mellitus and its Complications INTERDIAB (Bucharest, Romania, 5-7 March 2020), Filodiritto Editore – Proceedings, pp. 332-337
- Mihele, D., Pop, A., Dãrmãnescu, D., Cocu, F. (2008). Comparative studies regarding the influence of the esters and the ethanol amides of the fatty acids from extra-virgin Oleum Olivae on the experimental ulcers and on the gastric activity on rat Jul 2008 in Planta Medica, Vol. 74, pp. 995 – 1228
- Mihele, D., Darmanescu, D., Cocu, F., Pop, A.L., & Mihele, D. (2008). The anti-inflammatory action of the ethanol amides of the extra-virgin Oleum olivae fatty acids by experimental acute inflammation methods on rats, Romanian Biotechnological Letters, Vol. 13, (6) supplement, pp. 56 -61
- Bârcă, M., Manda, G., Ciobanu, A. M., Bălălău, C. Lupuleasa, D., Burcea Dragomiroiu, G. T. A. & Baconi, D. L. (2017). Immunomodulatory effects of methadone following methotrexate therapy in a rat model of arthritis. Farmacia, 65(3), 423-428
- Mititelu, M., Nicolescu, T. O., Moroșan, E., Pop, A. L., Nicolescu, F. (2020). Study of Hypolipemiant Activity of Some Mussel Extracts. Proceedings of 6 th International Conference on Interdisciplinary Management of Diabetes Mellitus and its Complications INTERDIAB (Bucharest, Romania, 5-7 March 2020), Filodiritto Editore – Proceedings, pp. 338-343
- Ioniță, A. C., Mititelu, M., Nicolescu, T. O., Nicolescu, F., Moroșan, E., Ozon, E. A., Ioniță, E. I. (2020). Research on the Hypolipidemic Action of Almond Oil and Almond Seeds. Proceedings of 6 th International Conference on Interdisciplinary Management of Diabetes Mellitus and its Complications INTERDIAB (Bucharest, Romania, 5-7 March 2020), Filodiritto Editore – Proceedings, pp. 326-331
- Ghica, M., Băncescu, I., Udeanu, D. I., Tăerel, A., Arsene, A. L., Anuța, V., Velescu, B. Șt., Gerghiceanu, F., Mititelu, M., Ioniță, A. C. (2020). Computational Approach for Small Data in Animal Models with Induced Metabolic Disorders, Proceedings of 6 th International Conference on Interdisciplinary Management of Diabetes Mellitus and its Complications INTERDIAB (Bucharest, Romania, 5-7 March 2020), Filodiritto Editore – Proceedings, pp. 98-106
- Buşuricu, F., Roşoiu, N. (2001). Etude des phospholipides de quelques especes d’invertébrés et de poissons de la mer noire, rapp. Comm. Int. Mer. Medit., 36, pp. 185
- Mititelu, M., Moroşan, E., Neacșu, S. M., Ioniţă, E. I. (2018). Research regarding the pollution degree from romanian Black Sea coast. Farmacia 66(6), pp. 1059-1063
- Mititelu, M., Ghica, M., Ioniţă, A. C., Moroşan, E. (2019). The influence of heavy metals contamination in soil on the composition of some wild edible mushrooms. Farmacia 67(3), pp. 398-404
- Mititelu, M., Moroşan, E., Iosif, M., Ioniţă, E. I. (2018). Analisys of quality of different types of honey from various sources, Proceedings of The Romanian National Congress of Pharmacy – 17th Edition, “21st Century Pharmacy – Between Intelligent Specialization and Social Responsibility”, Filodiritto Editore – Proceedings, pp. 84-87
- Marcolino, V. A., Zanin, G. M., Durrant, L. R., Benassi, M. T., Matioli, G. (2011). Interaction of Curcumin and Bixin with β-Cyclodextrin: Complexation Methods, Stability, and Applications in Food. Journal of Agricultural and Food Chemistry. 59 (7): 3348–57
- De Oliveira, V. E., Almeida, E. W. C., Castro, H. V., Edwards, H. G. M., Dos Santos, H. F., De Oliveira, L. F. C. (2011). Carotenoids and β-Cyclodextrin Inclusion Complexes: Raman Spectroscopy and Theoretical Investigation. The Journal of Physical Chemistry A. 115 (30): 8511–9
